KAPA Blood PCR Kits
 Eliminate the need for DNA extraction with the first DNA polymerase evolved for whole blood PCR.
Eliminate the need for DNA extraction with the first DNA polymerase evolved for whole blood PCR.
KAPA Blood DNA Polymerase is a second-generation enzyme derived through a process of molecular evolution and the first DNA polymerase engineered specifically for the amplification of DNA directly from whole blood. The enzyme is available in two optimized, easy-to-use 2x PCR mixes, which contain all components required for Whole Blood PCR, except primers and template (blood). Using KAPA Blood PCR Kits, DNA fragments may be amplified directly from reactions containing 1 – 20% (v/v) whole human blood without pretreatment of blood samples or DNA isolation, significantly reducing contamination risk, turnaround time and cost of genetic testing.
KAPA Blood PCR kits offer:
- Direct amplification from fresh or frozen whole blood, blood collected in EDTA tubes or on FTA® Elute Cards, “Guthrie cards” or filter paper.
- Eliminates the need for DNA extraction in genetic testing protocols for humans, other mammals and birds.
- Reduces contamination risk, turnaround time and cost of genetic testing.
- Compatible with existing workflows, standard protocols and routinely used detection methods.
KK7002 KAPA Blood PCR Mix A (500 x 25 µL rxns) KAPA Blood DNA Polymerase in a convenient 2X ReadyMix format. Mix A is the preferred formulation for assays employing fluorescent detection systems. Compatible with the Promega PowerPlex 16 System. KK7003 KAPA Blood PCR Mix B (500 x 25 µL rxns) KAPA Blood DNA Polymerase in a convenient 2X ReadyMix format. Mix B is the preferred formulation for assays using ethidium bromide detection and amplification of GC-rich targets directly from blood. KK7004 KAPA Blood PCR Mix A (1,000 x 25 µL rxns) KAPA Blood DNA Polymerase in a convenient 2X ReadyMix format. Mix A is the preferred formulation for assays employing fluorescent detection systems. Compatible with the Promega PowerPlex 16 System. KK7005 KAPA Blood PCR Mix B (1,000 x 25 µL rxns) KAPA Blood DNA Polymerase in a convenient 2X ReadyMix format. Mix B is the preferred formulation for assays using ethidium bromide detection and amplification of GC-rich targets directly from blood. Product Description
KAPA Blood PCR Kits are available in two optimized, easy-to-use 2x formats containing all PCR components except primers and template (whole blood).
KAPA Blood PCR Mix A is preferred for assays employing highly sensitive fluorescent detection systems. KAPA Blood Mix B is formulated for higher yields and GC-rich amplicons and is recommended when analysis is by agarose gel electrophoresis and ethidium bromide staining. Products generated with KAPA Blood PCR Mixes A or B may be analyzed by restriction endonuclease digestion, DNA sequencing or dHPLC analysis.
Product Applications
KAPA Blood PCR Kits have been validated for the direct amplification of DNA fragments from fresh or frozen whole blood, blood collected in EDTA anti-coagulant tubes, on FTA® Elute Cards, Whatman 903® Specimen Collection Paper (“Guthrie cards”) or regular filter paper. The optimal amount of blood per reaction depends on the species, amplicon type and application. KAPA Blood PCR Kits are recommended for the following:
- Genetic testing of humans, other mammals and birds.
- Amplicons with a GC content <65% up to 3.5 kb in length, and amplicons with GC content >65% up to 1 kb.
- Single amplicon or Multiplex PCR assays, using unlabelled or fluorescently labelled primers.
- Routinely used detection methods including restriction endonuclease digestion, agarose gel electrophoresis/ethidium bromide staining, fluorescent capillary electrophoresis, DNA sequencing and dHPLC analysis.
- KAPA Blood PCR Kits are ideally suited for paternity testing using the PowerPlex® 16 System (Promega).
Reduce contamination risk, turnaround time and cost of genetic testing
High-throughput genetic testing directly from blood has not been feasible with wild-type polymerases, due to the presence of multiple PCR inhibitors in whole blood. KAPA Blood DNA Polymerase was evolved in this environment and offers the ability to amplify DNA fragments directly from blood, without any pretreatment of blood samples or DNA isolation. This not only reduces the cost and turnaround time of genetic testing, but also significantly reduces the risk of sample cross-contamination associated with the need for DNA extraction.
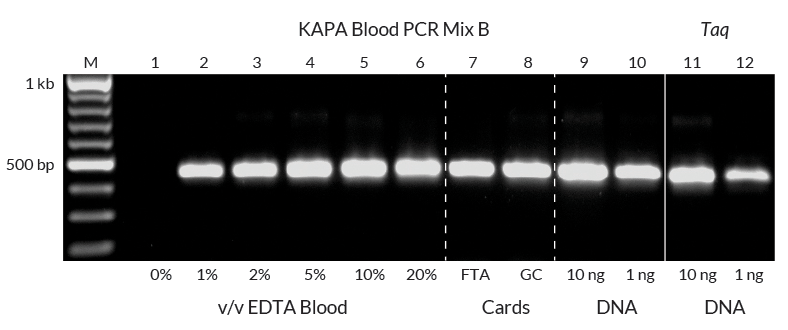
Amplification of a 459 bp fragment of exon 19 of the human Duchenne muscular dystrophy gene directly from whole human blood, using KAPA Blood PCR Mix B.
Reactions (50 µL) contained different amounts of blood (0 – 20% v/v, as indicated) from EDTA anticoagulant tubes stored at 4°C (lanes 1 – 6), or punches from an FTA® Elute Card (lane 7) or a “Guthrie card” (lane 8) as template. Control reactions, performed with 10 ng or 1 ng purified human genomic DNA as template and KAPA Blood PCR Mix B (lanes 9 and 10), or wild-type Taq polymerase (lanes 11 and 12) are included on the right. A standard 3-step cycling profile (35 cycles) with an initial denaturation of 5 min (95°C) and 1 min extension time per cycle was used in all reactions.
Compatibility with existing workflows and detection methods
KAPA Blood DNA Polymerase is supplied in two optimized, convenient 2x PCR Mixes, which are easily integrated with existing protocols and detection methods. Reaction products are centrifuged to recover amplified DNA from hematocyte debris.
Paternity testing using fluorescent capillary electrophoresis
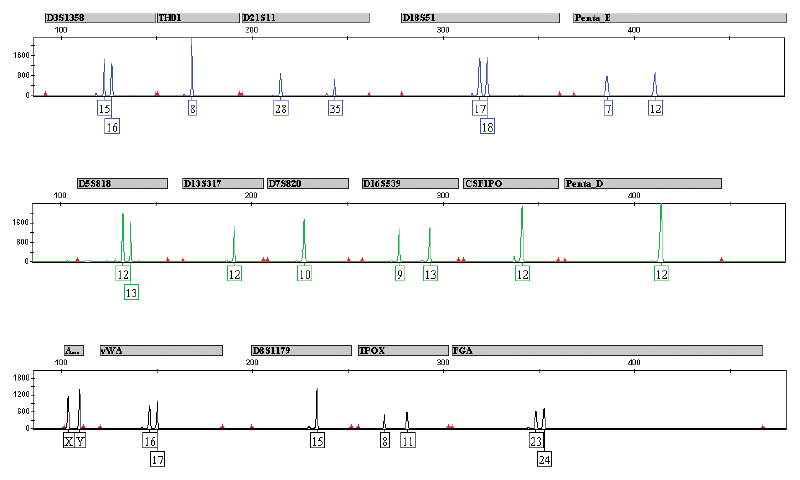
Typical result obtained in a PowerPlex16® (Promega Corporation) paternity test using KAPA Blood PCR Mix A, with blood collected on Whatman 903® Specimen Collection Paper (“Guthrie cards”) as template. The test is based on the amplification of 16 loci (15 STR loci and amelogenin) in a single Multiplex PCR using primers labelled with three different fluorophores. Reactions were set up and performed according to manufacturers’ recommendations. Reaction products were centrifuged and cleared supernatants were analyzed directly by fluorescent capillary electrophoresis using standard protocols. Image courtesy of Unistel Medical Laboratories.
Genetic testing based on RE digestion of DNA fragments amplified directly from whole blood
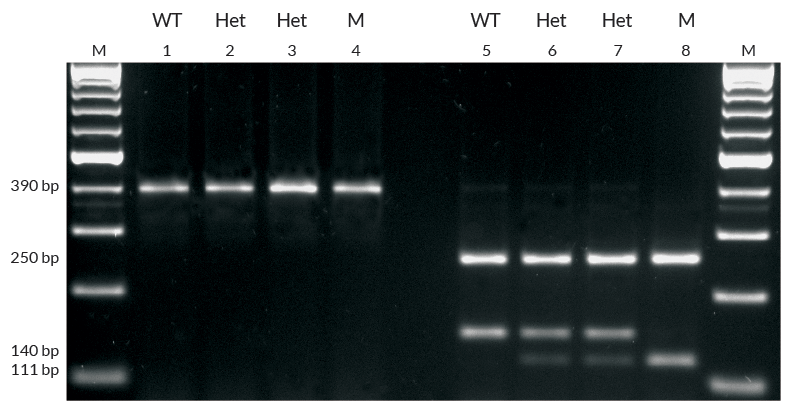
Incorporation of whole blood PCR in a PCR-based test for hereditary hemochromatosis (HH).
The amino acid mutation C282Y, associated with HH in >80% of homozygous individuals, is caused by a single nucleotide polymorphism (A845G) in the HFE gene. This SNP creates an additional site for restriction endonucleaseRsa I.A 390 bp fragment of the HFE gene, spanning amino acid C282, was amplified using KAPA Blood PCR Mix B in 50 µl reactions containing 10% v/v whole EDTA blood. Amplified DNA was recovered from cellular debris by centrifugation and digested directly with Rsa I. RE digestion products were electrophoresed in a 2.5% TBE-agarose gel and detected by ethidium bromide staining.
PCR products generated from the blood of homozygous wild-type (WT), homozygous mutant (M) and heterozygous (Het) individuals are shown on the left. RE digestion products (right) of the WT allele yields 2 fragments (250 bp + 140 bp), whereas the mutant allele yields 3 fragments (250 bp + 111 bp + 29 bp), of which the smallest is not detectable by ethidium bromide staining. Heterozygous individuals are identified by a digestion product consisting of the 250 bp, 140 bp and 111 bp fragments. Generated in collaboration with Unistel Medical Laboratories.
Direct amplification from the blood of non-human species
KAPA Blood PCR Kits may also be used for the direct amplification of DNA from the blood of non-human species, such as other mammals and birds, thereby offering cost and time savings in PCR-based veterinary testing. Blood from other species may be collected in the same manner as human blood. The optimal amount of blood used in a KAPA Blood PCR must be determined empirically for each species. With mouse blood, best results have been obtained with a lower concentration of blood in the PCR (0.1 – 5% v/v) than recommended for humans. For bird species with nucleated erythrocytes, blood should be diluted to more closely approximate the DNA concentration in the same volume of human blood.
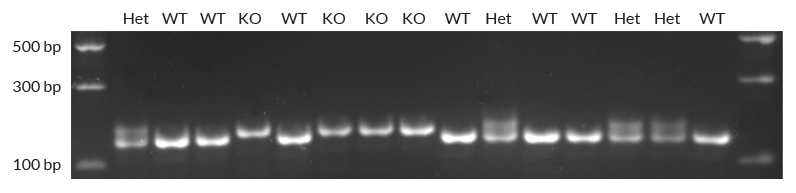
High-throughput genotyping of mice used in investigations into the relationship between metallothionein (MT) expression and mitochondrial function. MT knockout mice carry a short insertion in the MT gene on both alleles, which results in the absence of functional metallothionein. To distinguish mice carrying the knockout mutation on one or both alleles from homozygous wild-type mice, a fragment of the MT gene is amplified directly from mouse blood collected on “Guthrie cards”, using KAPA Blood PCR Kit A. A standard 3-step cycling protocol (30 cycles) with an initial denaturation time of 10 min (95°C) was employed. PCR products were cleared by centrifugation, electrophoresed in a 2.5% agarose gel and detected by ethidium bromide staining.
A single larger band corresponds to a homozygous knockout mouse (KO), whereas a single smaller band corresponds to the homozygous wild-type(WT). Heterozygotes (Het) yield a double band representative of both the KO and WT alleles. Image courtesy of North-West University.